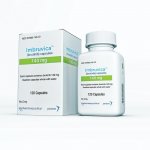
Italian pharmaceutical company Alfasigma is set to acquire Intercept Pharmaceuticals in an approximately $800 million all-cash deal. The acquisition will bolster Alfasigma’s presence in the U.S. market and expand its gastrointestinal and hepatology portfolio. Intercept’s obeticholic acid (OCA), marketed as Ocaliva, will become part of Alfasigma’s offerings. Ocaliva is the only approved second-line treatment for primary biliary cholangitis (PBC). This transaction follows Intercept’s decision to cancel its non-alcoholic steatohepatitis (NASH) program and lay off staff after the FDA rejected its NASH treatment in June 2023. The deal is expected to close by the end of 2023.
Italian pharma Alfasigma S.p.A is buying Intercept Pharmaceuticals, a biotech specializing in rare and serious liver diseases, in a roughly $800 million cash deal, the companies announced Tuesday.
The “anticipated transaction” at $19 per share “will materially expand Alfasigma’s gastrointestinal and hepatology portfolio and its presence in the U.S. market,” the companies said in a press release. The companies also noted the “purchase price represents a premium of 82% to Intercept’s closing stock price on September 25, 2023.”
The deal will bring Intercept’s obeticholic acid (OCA), marketed as Ocaliva, into Alfasigma’s portfolio. The farnesoid X receptor is the only approved second-line treatment for primary biliary cholingitis (PBA), in which the body’s bile ducts in the liver are slowly destroyed, and has “experienced double-digit year-over-year growth,” the companies noted.
Want to publish your own articles on DistilINFO Publications?
Send us an email, we will get in touch with you.
The news comes just months after Intercept’s second push for regulatory approval of its treatment for non-alcoholic steatohepatitis (NASH) came to an end. In June 2023, the FDA rejected the company’s OCA tablets as a potential treatment for NASH patients with pre-cirrhotic liver fibrosis, saying the drug “could not be approved in its current form and would require at least the completion of the Outcomes part of its Phase III trial.”
The next day, Intercept announced it was cancelling its NASH program and also laying off about a third of its workforce. The plan was to increase investment in Ocaliva and focus R&D on a combination of OCA and bezafibrate to treat PBA. The cuts were anticipated to save $140 million per year, allowing the company to potentially become profitable by 2024.
The initial rejection in November 2019 came after the regulatory determined the available data from Intercept “was not enough to establish that its benefits would outweigh its risks.”
While the purchase announcement has bolstered Intercept’s stock value, with Reuters reporting a 77.5% jump to $18.53 in Tuesday morning trading, the wire service also noted that price is far from the 2014 peak of $445 per share. The deal is expected to close by the end of 2023.
It was fully funded by Alfasigma’s existing cash on hand and corporate credit facilities, the companies noted in their announcement, adding that the deal was approved unanimously by both boards of directors.
Alfasigma is a privately owned company, with over 75% of the ownership in the hands of the founder Marino Golinelli’s family. Once the purchase is complete, Intercept will also become private, the companies said. “Upon completion of the transaction, Intercept’s common stock will no longer be publicly listed.”
Source: BioSpace