- Ed Francis, Global Managing Partner, Health and Life Sciences (U.S.)
- Debbie Cooper, Global Managing Partner, Drug Development and Regulatory (U.K.)
- Contributions from Ellen Vanbuskirk, Associate Partner, healthcare strategist (U.S.)
- Vishal Singal, Associate Partner, AI, Analytics and Data (U.S.)
The COVID-19 virus has had a devastating impact on our communities, personal health and economic vitality. For the week ending March 21 the John Hopkins tracking site reported more than 315,000 cases and more than 13,500 deaths. 1 Appropriately, all major governments have been faced with taking draconian efforts to flatten the infection curve and allow our healthcare system to keep pace with the number of ill patients. However, the spread continues to increase and our healthcare providers are facing shortages of critical medical equipment and supplies, including ventilators, masks, testing swabs and protective gear.
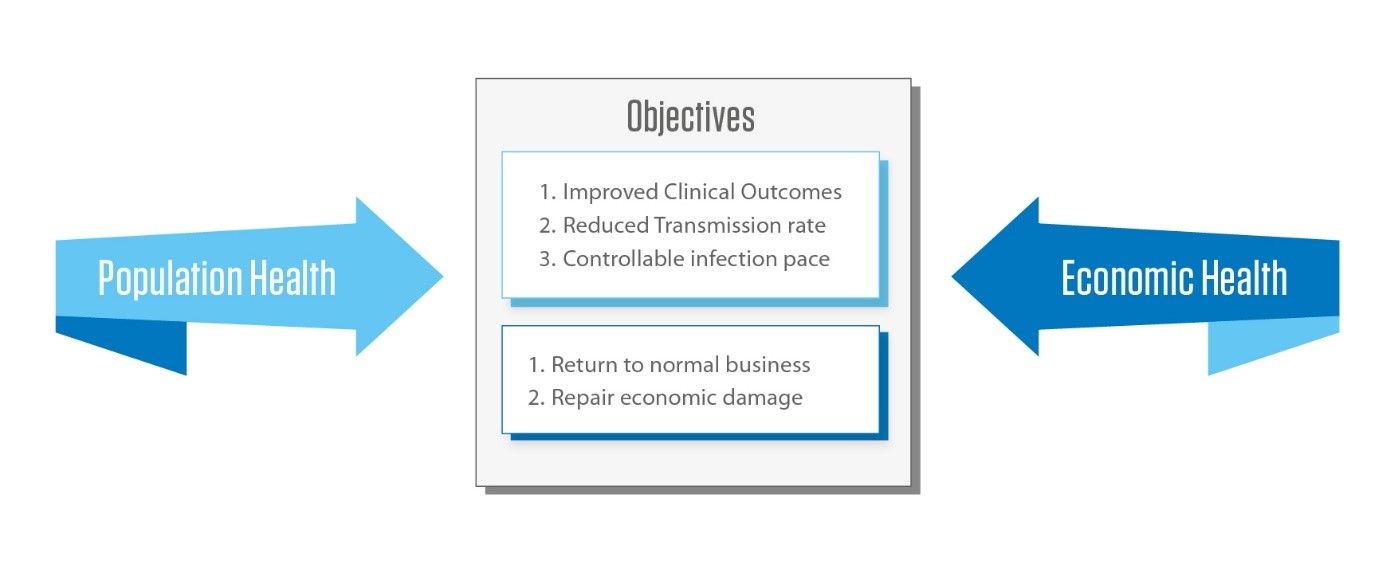
The challenges faced goes beyond mitigating the spread of this virus by flattening the curve to ensure we do not overwhelm our health system. It is also an economic challenge that must be considered alongside the medical and scientific expertise being put toward managing the pandemic.
Goldman Sachs revised its 2020 economic forecast predicting a 6% decline in Q1, a 24% decline in Q2, but then rebounding by 12% in Q3 and 10% in Q4 for a decline of 3.8% in full-year growth. 2 As illustrated above, the significant challenge of meeting the needs of population health seem at odds with ensuring that normal economic activity resumes as quickly as possible.
Balancing Desired Outcomes
Want to publish your own articles on DistilINFO Publications?
Send us an email, we will get in touch with you.
Following are 7 predictions and 8 bold actions that offer perspectives on how to address the needs of population health while speeding the return to normal business activity.
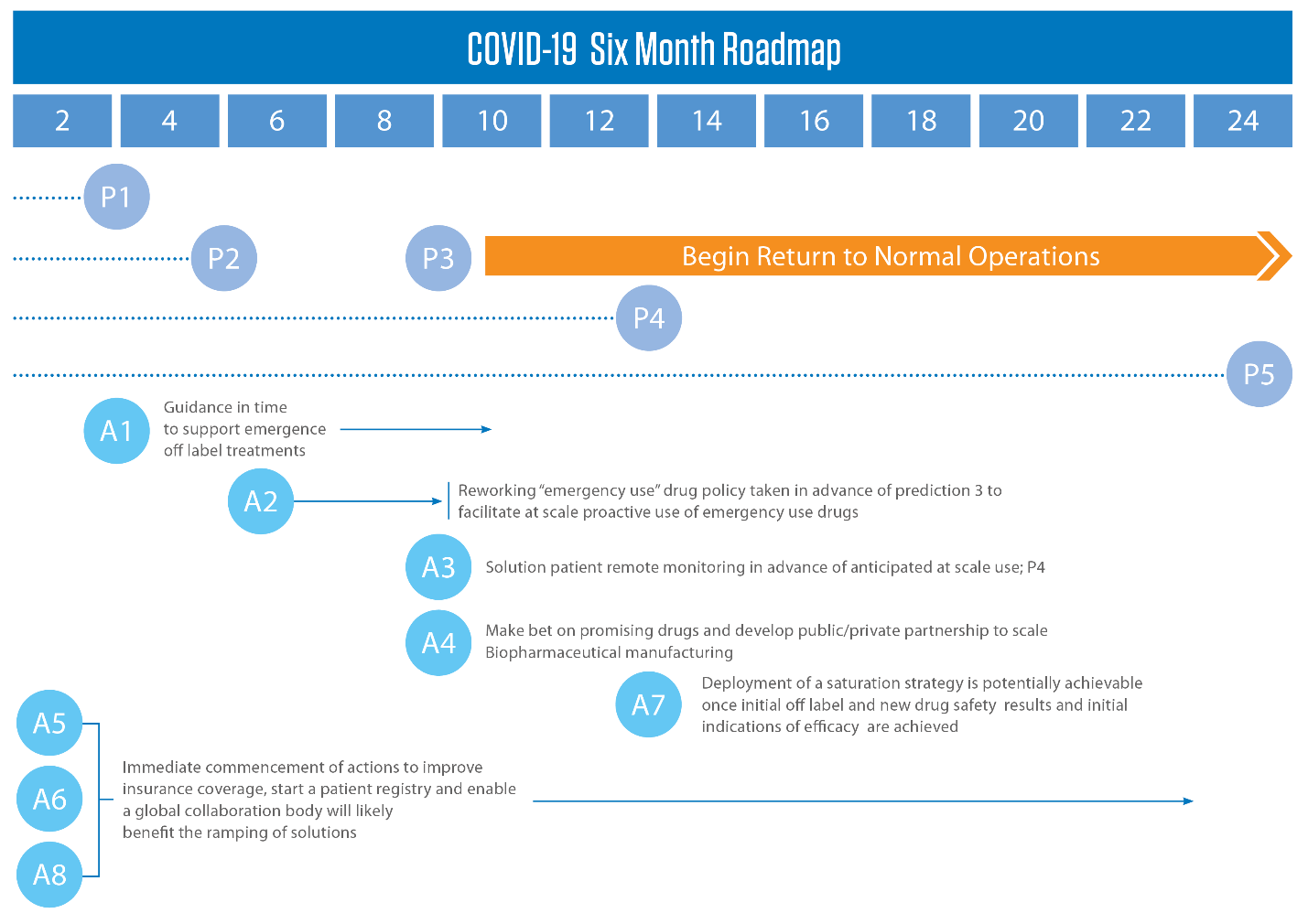
The 7 predictions and 8 actions are illustrated in the above potential 6-month roadmap. The illustration links the proposed actions (‘A’ circles on bottom half) to the predictions (‘P’ circles in top half) to show how they can work together to accelerate improvement in population health and return to normal business activity.
Prediction 1: Within one week there will be recognized treatment leveraging existing drugs off-label. Just last week Australian doctors reported that a combination therapy of HIV drugs lopinavir/ritonavir paired with anti-malaria drug Chloroquine has had a significant positive effect on COVID-19 patients. Both the federal and NY state governments have recently put resources behind a drug combination of Hydroxychloroquine and an antibiotic Zithromax by expediting a reported 10,000 doses for use in [seriously] ill COVDI-19 patients. 3
Finally, Chloroquine by itself is already being recognized as somewhat effective against COVID-19. 4WHO have launched a global megatrial of the four most promising coronavirus treatments. The study, which could include many thousands of patients in dozens of countries, has been designed to be as simple as possible so that even hospitals overwhelmed by an onslaught of COVID-19 patients can participate.
Prediction 2: Within four weeks there will be numerous treatments leveraging existing drugs off-label. Already, there are a wide range of drug and drug combinations that are gaining traction. Gilead’s anti-viral drug Remdesivir and Takeda’s plasma derived therapy approach (TAK888).5 In addition, just last week a group of researchers “identified 31 existing broad-spectrum antiviral agents (BSAAs) that may be potential candidates for repurposing against the infection”.6
Prediction 3: Within eight weeks we will begin to see a mitigating effect from weather. Based on current news stories, it is unclear whether summer weather will diminish the frequency of COVID-19 infections that occur in the same manner it does for the seasonal flu virus. However, there appears to be substantial evidence suggesting that warm weather will have a mitigating effect on the transmission of the virus. The basis of evidence centers on analog virus studies such as SARS, correlation analysis of the current spread of COVID-19 and laboratory analysis of temperature effect on COVID-19.7 It is true that much of the evidence has not been peer reviewed and in the case the COVID-19 temperature analysis, the source data is questionable particularly given detection challenges and lack of testing. Additionally, there has been an increase in the number of cases in hotter8 climates such as Australia but much of them have been imported from Europe or United States. The spread of disease in countries such as Cambodia, Philippines has been at rate slower than mass outbreak seen in other countries. Despite these issues, the evidence in aggregate seems to indicate that summer weather will mitigate the virus’ transmission rate.
Prediction 4: Within 12 weeks initial safety results will become available from new drugs and the FDA will approve some drugs for emergency and humanitarian use. It was announced that a vaccine produced by Moderna began Phase 1 clinical trials for the first COVID-19 vaccine on March 16. The intent of this Phase 1 study is to test safety and immunogenicity of three dose levels of mRNA-1273 (25, 100, 250 μg) to be given on a two-dose vaccination schedule, with a gap of 28 days.9
Beyond the Moderna trial there are dozens of clinical trials in place with more to come. The release of safety results and initial efficacy evidence will be fast coming offering more opportunities to consider “emergency” or “compassionate” use opportunities. It is predicted that there will be enough safety data for regulators to seriously consider leveraging emergency use” and “compassionate use” clauses within regulatory law to administer the most promising vaccines to at-risk populations and perhaps beyond.
Prediction 5: Within 24 weeks some COVID-19 Vaccines will be approved for use at scale through existing or new humanitarian-use laws and policies. The challenge with this prediction is it is questionable whether regulatory law governing drug emergency and compassionate use can be applied proactively when a potential patient has not been infected. This crisis will forever change how we deal with pandemics and by necessity through either policy change or a broader interpretation of existing law it is predicted that vaccines will be approved for use at scale.
Prediction 6: Within 12 months, clinical trials will never be the same. The U.S. started its first clinical trial on March 16 and China began its first on March 18.10 Already, we are seeing streamlined and expedited clinical trial processes with heightened collaboration between pharmaceutical companies and the FDA. It is becoming more evident that a more agile clinical trial process is strategically important to human health now and to help mitigate and manage future global pandemics.
It may result in Health Authorities agreeing to accept approvals from agencies outside of their jurisdiction as an accelerator secure supply in exceptional circumstances, similar to the Mutually Recognized Procedure (MRP) in the EU. In addition, it recognizes that our current processes are woefully inadequate to deal with an emergent pandemic, resulting in greater collaboration and competition to create an effective new process.
Prediction 7: By 2022 biopharmaceutical manufacturing will become a strategic asset. As Chloroquine has emerged as a possible treatment for COVID-19 the world is facing significant shortages of this drug as well as critical medical equipment and supplies. UK pharmacies have reportedly sold out of the drug, and the UK government has banned export of three drugs to other countries11. If the previous predictions come true, it is easy to imagine a need to suddenly produce billions of doses of, in many cases, complex biopharmaceuticals. The global distribution of a limited supply could easily be confined to country of origin regardless of ownership. It seems likely that the world will learn from this and view bio-pharmaceutical manufacturing as a strategic asset and seek more in-country manufacturing.
Action 1: Government health agencies should take the lead in advising on current drug off-label use and new drug emergency and compassionate use. Under the current US system, doctors are on their own to make difficult decisions about off-label drug use or to provide advice on using new drugs while simultaneously treating a surge in new patients. The providers are left to take the burden of tort risk when prescribing off-label drug use and some won’t do it leaving the patient in a vulnerable position. Central advice and the FDA, CDC, NIH, EMA, associations and others are well positioned to evaluate information and provide guidance would ensure consistent decision making. Additionally, consideration of a law or policy to protect providers who follow reputable advice and adhere to prescribed protocols could enable scaling the use of off-label and emergency use clinical trial drugs.
Action 2: Rework unapproved drug law and policy: To support the use of an unapproved preemptive drug (e.g., vaccine), there likely needs to be a rewrite of the U.S. emergency and compassionate use policy and law to ensure that proactive drugs such as Vaccines can be allowed. The law should enable all the treatments to be used as long as informed consent is obtained from patients and protocols are followed, with close monitoring and reporting of any adverse events as it was done in the case of Ebola12 across the world. Other global economies are likely to follow with statute changes.
Action 3: Scale remote patient monitoring capabilities: Remote monitoring of patients using existing wearable technologies can be an important capability in supporting the scaled use of off-label drugs or emergency and compassionate use of drugs. It can also be used to provide reassurance and support to vulnerable patients and their caregivers without the need to visit a healthcare facility reducing the likelihood of exposure to COVID-19 or other similar infectious diseases. It can be used as a mechanism to help mitigate the risk of using unapproved drugs. In addition, remote monitoring is already gaining widespread use in clinical trials today and should therefore be an easy action to take.
Action 4: Scale Biopharmaceutical manufacturing capacity: Should suitable off-label and emergency-use treatments be found for COVID-19, it is likely that our current drug manufacturing capacity will be insufficient to meet the needs of the U.S. population much less a global population. This will be particularly true with biopharmaceuticals manufacturing which is much more complex than the manufacture of traditional, small molecule drugs.
To further complicate the challenge, most biopharmaceutical manufacturing is proprietary and either patented or considered a trade secret. It is difficult to imagine many companies willing to risk investing hundreds of millions of dollars to scale the manufacture of a drug that has not been FDA approved for a COVID-19 indication. However, the Federal Government can start to play a pivotal role now in helping proactively scale global manufacturing for drugs that have high potential for solving the crisis. If government can help minimize the risk through funding, guarantees, etc., it will be possible to dramatically improve the speed with which we collectively respond to the crisis by ramping up our ability to manufacture the needed medicines and treatments
Action 5: Global patient registry of cured patients: Many will question breach of privacy and others will site the lack of testing as a major impediment to creating a registry, but there are several compelling reasons why this could significantly help fight the crisis:
- One potential treatment is plasma therapy, such the one being tested by Takeda Pharmaceuticals, which will rely on plasma from recovered patients. The ability to quickly identify these patients (using digital technologies available today) and incent the provision of blood could dramatically help scale this treatment option.
- Better data can help model disease transmission and forecast herd immunity, which is when a sufficient number of the population has developed immunity such that transmission of the virus is stopped. Understanding this information in conjunction with other preemptive measure can help better identify at-risk communities and enable better targeting of vaccine deployment and other solutions.
- Traceability of people who have had COVID-19, allows a core of “immune” people to return to work, enabling the economy to stabilize more quickly and to potentially provide second-line support to the healthcare systems.
- Inclusion of this data in genomic profiling tools alongside additional diagnostic viral testing will ensure that mutations and the longer-term effects of the virus can be understood. Some virus’ remain dormant in the system and can cause long-term impact on health when the reoccur, for example Epstein-Barr, Cytomegalovirus, HIV.13 Payer/Providers can also use this information for predictive risk profiling.
- Improvement in the time taken to provide “Point of Testing”, results immediately will result in much improved patient outcomes. Testing times have already reduced from days to minutes. 14The FDA approved a test that can detect the coronavirus within 45 minutes and can be processed without training.
Action 6: Facilitate insurance approval for experimental and off-label use drugs: Many people have experienced challenges getting insurance approvals for off-label drug use and experimental drugs. Large-scale use of unapproved drugs as done in the clinical trial setting is often paid for by the pharmaceutical company. If the use of off-label and experimental drugs increases, the current models of pricing and payment will break down. There will also be challenges for the uninsured in terms of affordability and access. One option is for, the US government to play a short-term role in brokering a public-private solution to this problem.
Action 7: Deploy a saturation strategy to increase public immunity: In a recent interview, Larry Brilliant, chairman of the board for Ending Pandemics (endingpandemics.org) and a former epidemiologist who helped eradicate smallpox 14 years ago, discussed the potential of deploying a saturation strategy to facilitate public immunity against the virus.15 Saturation is defined as proactively administering a drug in a high enough percentage within a population to create a herd immunity; usually at levels of 70-80%. Brilliant has model this for influenza by using Tamiflu and has shown that his approach works., Given the serious nature of COVID-19 perhaps this is a recommendation worth serious consideration and only the government is likely to be able to facilitate.
Action 8: Establishment of global governance body: We are already beginning to see nationalistic interests emerge to protect local citizens as well as efforts to cooperate globally. A good example of global cooperation is as part of International Coalition of Medicines Regulatory Authorities (ICMRA) global regulators are working together to map out data requirements for COVID-19 data requirements.16 Nevertheless, there are lot of challenges ahead with regard to optimizing global manufacturing capacity, ensuring free flow of clinical information, protection of IP rights, distribution of potentially scarce drugs and many others. Although nationalistic and individualistic tendencies in the midst of a life and death pandemic cannot be avoided, the establishment of a global governance or collaboration body can certainly help bring a level of cooperation and collaboration that will benefit the world’s population and a faster recovery for everyone.
This unprecedented lifetime event has highlighted the best of innovation and resilience. Drug approval and deployment evolution will be critical to arresting the current pandemic. It will involve applying agile approaches; properly assessing risk and reward and making big bets to dramatically shorten the time to find and scale new therapies and drugs to address the current crisis and healthcare will never be the same again.
2 https://finance.yahoo.com/news/goldman-forecasts-stunning-drop-economic-230052586.html
3 https://www.voanews.com/science-health/coronavirus-outbreak/ny-state-begin-testing-malaria-drug-covid-19-battle
4 https://www.sciencemag.org/news/2020/03/who-launches-global-megatrial-four-most-promising-coronavirus-treatments
5 https://www.thestreet.com/investing/takeda-eyes-plasma-therapy-for-covid-19-patients
6 https://www.genengnews.com/news/coronavirus-treatment-could-lie-in-existing-drugs/
7 https://www.msn.com/en-us/news/weather/higher-temperatures-affect-survival-of-new-coronavirus-pathologist-says/ar-BB119VQo
8 https://www.washingtonpost.com/weather/2020/03/24/warm-humid-weather-coronavirus/
9 https://www.pharmaceutical-business-review.com/news/moderna-mrna-1273-coronavirus-trial/
10 https://www.bioworld.com/articles/433791-china-approves-first-homegrown-covid-19-vaccine-to-enter-clinical-trials
11 https://www.dailymail.co.uk/news/article-8134125/Boris-Johnson-announces-start-coronavirus-clinical-trials-Britain.html
12 https://www.who.int/ebola/drc-2018/treatments-approved-for-compassionate-use/en/
13 https://academic.oup.com/cid/article/31/3/739/297506
14 https://www.businessinsider.com/fda-approves-coronavirus-test-that-can-diagnose-in-45-minutes-2020-3
15 https://arstechnica.com/science/2020/03/the-doctor-who-helped-defeat-smallpox-explains-whats-coming
16 https://www.ema.europa.eu/en/news/global-regulators-map-out-data-requirements-phase-1-covid-19-vaccine-trials







