A UK-based health technology firm has received 510(K) clearance from the US Food and Drug Administration (FDA) for its AI-powered decision support system designed for the early detection of cardiovascular disease.
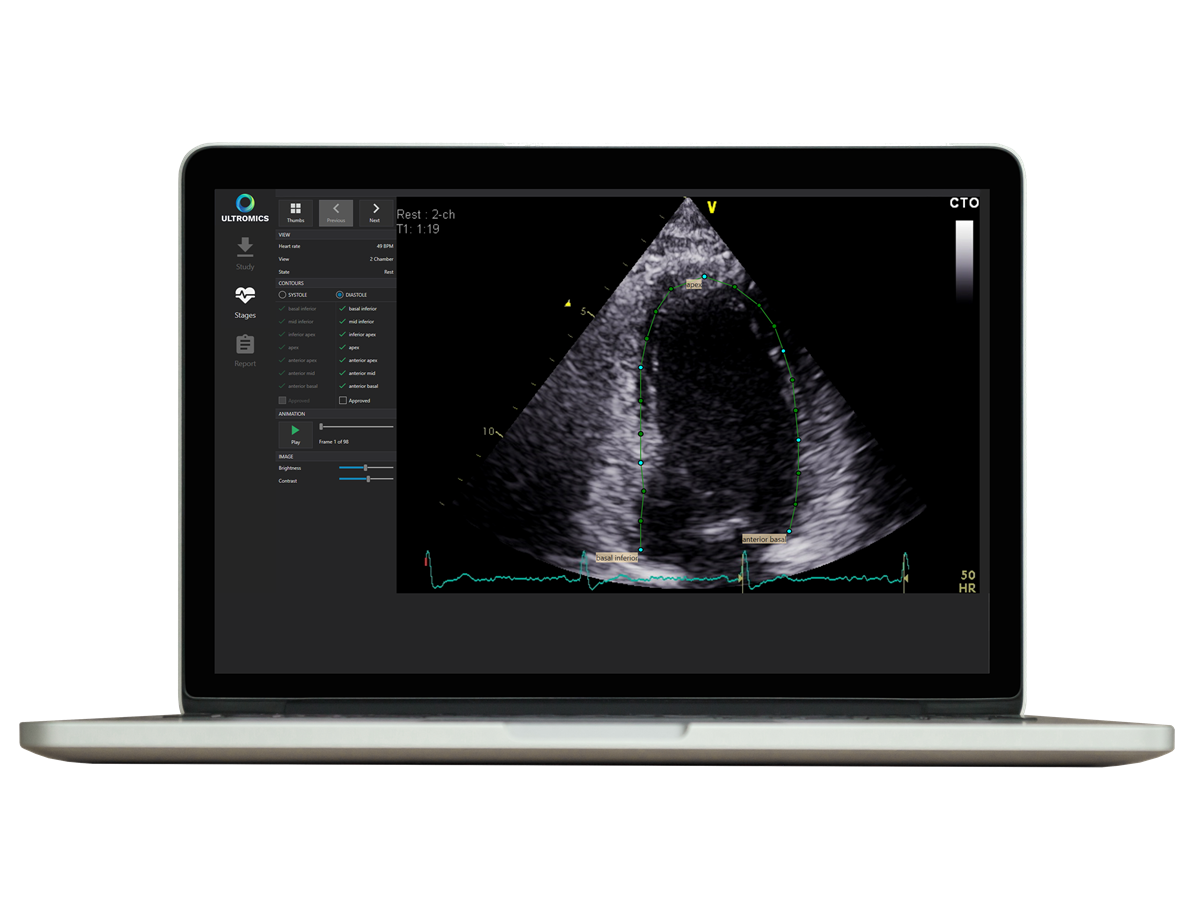
Ultromics, a UK company specialising in the application of artificial intelligence (AI) to echocardiography (an ultrasound of the heart), developed the image analysis system – called EchoGo Core – which aims to help identify cardiovascular disease in patients at an early stage. This is achieved by applying AI to the analysis and quantification of ultrasound-based heart scans.
According to Ultromics, the regulatory clearance is a major milestone for the company and enables the business to make EchoGo available to clinicians and healthcare providers in the US.
“This is an incredibly exciting step towards the future of healthcare,” said Ross Upton, founder and CEO of Ultromics. “EchoGo will help clinicians make more accurate and informed decisions to improve patient care delivery. It’s truly a watershed moment for Ultromics.”
Want to publish your own articles on DistilINFO Publications?
Send us an email, we will get in touch with you.
There are approximately 50 million heart scans conducted a year in the US, with cardiovascular disease the number one killer worldwide. According to a report from the British Heart Foundation (BHF), published in August this year, 7.4 million people in the UK are living with a heart or circulatory disease.
Traditionally, echocardiography has relied on the expert eye of clinicians, with years of experience, measuring the anatomical structures and identifying the disease, a potentially time-consuming and highly variable process.
However, by automating the process and applying its AI analysis to look in greater detail at the scans, the company said EchoGo enables clinicians to interpret echocardiograms efficiently and accurately and assists in their decision-making.

EchoGo uses AI to calculate left ventricular ejection fraction (EF), the most frequently used measurement of heart function, left ventricular volumes (LV). It is also the first AI application to measure cardiac strain.
“Strain has shown to be very valuable in cardiovascular diagnostics and has been demonstrated in published studies to be linked with earlier detection of disease and improved patient outcomes,” Upton said. “Ultromics’ will be the first to use artificial intelligence for automated strain analysis which is applicable to 60 million scans per year.”
He added: “Crucially, strain is also becoming reimbursable from January 2020 in the US and EchoGo allows clinicians across a wide range of experience levels to rapidly obtain accurate and repeatable calculations of strain parameters, assisting them in interpretation of echocardiograms.
“We have more developments planned in 2020, including EchoGo Pro. It will be the first AI system able to predict cardiac disease from echocardiography. Our goal is to improve patient outcomes through earlier detection of cardiac disease.”
Ultromics currently has partnerships with leading cardiology clinical centres in the US and 30 NHS centres in the UK. Through these collaborations, it is continuously optimising its scanning algorithms by analysing the archive of scans held by these centres.
According to the firm, the pipeline of AI algorithms underpinning EchoGo was built using thousands of images from a carefully curated dataset, from one of the largest echo studies of its kind.
Source: E&T








