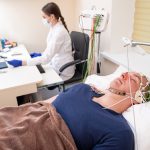
Medical experts are constantly being charged with the finding of new cures and innovative medical treatments that can save lives, yet without the eager participation of everyday patients in clinical trials it can be difficult or even downright impossible to achieve these breakthroughs in a time and cost-effective manner. Physicians who are concerned about the future of the healthcare industry are increasingly waking up and realizing that they need to help guide patients through clinical trials if they want to public to actively participate in the discovery of next-generation medical breakthroughs.
Nevertheless, many physicians don’t know where to begin when it comes to guiding patients through this arduous and frightening process. Here’s how physicians can help guide patients through clinical trials more effectively, and what you can do to ensure the entire thing is a patient-centered affair.
Enrollment targets aren’t being met
Anyone involved in the clinical trials process can tell you that enrollment targets aren’t being met around the nation right now as physicians are finding it challenging to lure in enough patients eager enough to go along with the process. There are many reasons that clinical trials struggle to drum up enough patient interest in order to succeed, but recent research has elucidated that there could be some structural barriers that are preventing clinical trials from reaching their full potential.
Want to publish your own articles on DistilINFO Publications?
Send us an email, we will get in touch with you.
According to researchers at the Fred Hutchinson Cancer Research Center, modern clinical trials are suffering from lackluster participation rates because structural barriers keep patients away from the process from the very get-go. Their new study argues that researchers who fail to drum up active participation in their clinical trials could see the results of such trials grow to be non-relevant as they fail to produce timely results. The researchers argue that a number of steps must be taken to eradicate these structural barriers, including offering clinical trials in more locations and updating patient criteria to fit modern demographics.
Physicians that want to help guide their patients through clinical trials should make themselves readily available whenever those patients are having serious concerns about the process. Medical research can be time-consuming and intimidating, not to mention a little uncomfortable, so trying to personalize the process and remind your patient that they’re a valued human being instead of a simple lab rat is important. Doctors and nurses who don’t go out of their way to calm patients and be a comforting presence throughout the clinical trials process will seldom see positive results develop over time.
You need to make the process patient-centric
Making your clinical trials patient-centric may seem obvious, but far too many medical professionals are too absorbed in their research to realize that the human element can’t be put on the sidelines if you want to succeed. Despite efforts to automate research or make paid clinical studies as dispassionate and scientific as possible, clinical trials are still fundamentally dependent on the good character of those who volunteer for them, making it important that you don’t dehumanize this process.
You should review five useful steps for making the process as patient-centric as possible, beginning with the creation of extensive patient profiles that go beyond traditional boundaries to tell you plenty about the emotional profile and personal preferences of the patients in question. You want to develop a serious, real connection to these patients over time, as that’s the only effective way to garner meaningful results from your lengthy and complex trials.
It’s also crucial to make the entire process of informed consent as clear as possible to patients. Physicians who don’t thoroughly understand the process of informed consent are likely to have their clinical trials burst into flames before they produce meaningful results that can be legally used, so make sure patients are familiar with their bill of rights before you start probing or poking them with needles. Whatever else you do, don’t forget that this process can be frightening and unfamiliar for patients who are foraying into clinical trials for the first time, so try to be a happy and warm person they can reach out to at any moment.
Making yourself constantly available and stressing informed consent is the only way that physicians can help guide patients through clinical trials with success. Before long, you’ll realize that a patient-centric process is the only surefire way to approach clinical trials.
Date: May 07, 2019
Source: Science World Report