1. Abbott

[Image from Abbott]
Abbott’s FreeStyle Libre System is a glucose measuring system that eliminates the need for finger sticks or finger stick calibrations. It measures glucose levels through a small sensor that is applied to the back of a user’s upper arm. The sensor is the size of two stacked quarters and gives real-time readings for up to 10 days, according to the company. The sensor is also able to read glucose levels through clothes.
Each scan provides three pieces of data, including real-time glucose results, an eight hour historical trend and a trend arrow showing the direction their glucose is headed. The accompanying touchscreen reader can hold up to 90 days worth of data to allow people to track glucose levels over time.
2. Boston Scientific

[Image from Boston Scientific]
Want to publish your own articles on DistilINFO Publications?
Send us an email, we will get in touch with you.
The Axios stent and electrocautery enhanced delivery system is designed to offer therapeutic endoscopists to safely and accurately access a pancreatic fluid collection from the gastrointestinal tract to place an Axios stent. The system uses a cautery-enabled access catheter with the therapeutic Axios stent for an exchange-free procedure.
The Axios catheter is energized and deployed through tissue walls and into target structures under EUS guidance. The catheter sheath is retracted which allows the stent to be deployed. Then, the delivery system is withdrawn.
3. Boston Scientific
[Image from Boston Scientific]
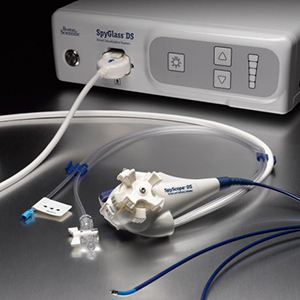
Boston Scientific’s SpyGlass is a direct visualization system that is used for cholangiopancreatoscopy. It is designed to optimize procedural efficiency and productivity because of its improved ease of setup, ease of use and image quality, according to the company.
It features a single-use scope that eliminates optical probe reprocessing and reduces risk of cross-contamination and image degradation over multiple uses. The integrated digital sensor features higher resolution image quality and 60% wider field of view for efficient evaluation of ducts with consistent articulation direction and accessory exit point. It also has a dedicated irrigation and aspiration connection that enables greater physician control to efficiently clear field of view. The tapered catheter tip has an atraumatic design that can assist with cannulation and traversing tight strictures, according to the company.
SpyGlass offers enhanced visualization and access for diagnostic and therapeutic applications like biliary system cancer, pancreatic cancer, bills duct cancer, gallbladder cancer, intraductal pancreatic mucinous tumor, intraductal papillary mucinous neoplasms of the pancreas, gallstones, primary sclerosis cholangitis, pancreatitis, pancreatic stones/debris, biliary papillomatosis and indeterminate pancreatic strictures and masses.
4. Boston Scientific
[Image from Boston Scientific]
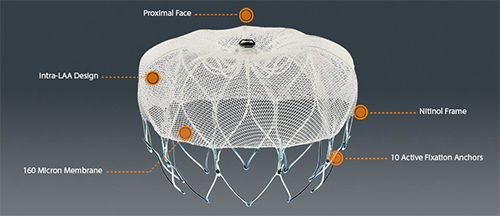
The Watchman from Boston Scientific is a left atrial appendage closure device that helps reduce stroke risk. It is an alternative to long-term warfarin therapy in patients with non-valvular atrial fibrillation.
Boston Scientific’s Watchman device is delivered through a transfermoral approach that is designed to close the left atrial appendage to prevent migration of blood clots, according to the company. Because it prevents blood clots, the device reduces the risk of stroke and systemic embolism.
5. Brainscope Company

[Image from Brainscope]
BrainScope’s BrainScope One system is an easy-to-use, non-invasive, handheld platform that allows physicians to assess head injures at point-of-care.
The device is FDA-cleared and is based off of EEG technology. BrainScope One is non-invasive and painless and can be used on patients who are aged 18-85 years old within three days of a head injury. It measure and interprets brain activity and neurocognitive function to help physicians make clinical diagnoses.
6. Foundation Medicine

FoundationOne CDx
foundation-medicine
FoundationOne CDx is an FDA-approved broad companion diagnostic that is used for solid tumors, including colorectal, breast, ovarian and melanoma. It is designed to give physicians clinically actionable information, according to the company.
Each test has microsatellite instability and tumor mutational burden to help make immunotherapy decisions.
7. Integra Lifesciences

Omnigraft Dermal Regeneration Matrix
The Omnigraft Dermal Regeneration Matrix from Integra Lifesciences is an advanced bilayer dermal regeneration matrix that treats diabetic foot ulcers. It is the only FDA-approved product that can regenerate native dermal tissue, according to the company.
The patch has a built-in silicone layer that serves as a temporary epidermal layer and provides immediate coverage for wound protection. It can be removed between two and three weeks. Under the silicone layer is a collagen/chrondroitin-6-sulfate matrix that acts as a dermal replacement layer. The Omnigraft features a bioengineered scaffold that promotes dermal regeneration and is designed with a controlled porosity and degradation rate.
The Omnigraft Dermal Regeneration Matrix is designed to be used for treating partial and full thickness neuropathic diabetic foot ulcers that are more than six weeks old with no capsule, tendon or bone showing.
8. Koning Corporation

[Image from Koning]
The KBCT from Koning is a breast imaging device. It offers high-contrast, real 3D images of the breast with spatial resolution and no painful compression in a 10-second exposure with radiation levels that are the same as normal mammograms.
The real 3D image features isotropic images in a 3D display to allow clinicians to evaluate tissue from any angle while eliminating overlapping structures. KBCT’s 3D scan is non-contrast, contrast enhanced and shows blood vessels and calcifications.
9. Medtronic

Implantable system for Remodulin
Medtronic’s implantable system for Remodulin is designed to treat patients who have pulmonary arterial hypertension. The system features the company’s SynchroMed II implantable drug infusion pump with a newly developed intravascular catheter that deliver Remodulin intravenously to patients who have previously received the drug through an external infusion pump.
Medtronic announced on July 31 that the system received FDA approval.
10. Otsuka Pharmaceutical

Abilify MyCite
Abilify MyCite is a prescription medication with a sensor inside that is designed to treat a number of conditions, including schizophrenia, bipolar I disorder and major depression disorder.
The Abilify MyCite system tracks if the medication has been taken. Usually ingestions can be detected with 30 minutes, but it could take up to two hours for a smartphone app or web portal to detect if the medication has been taken.
Date: August 6, 2018
Source: MedicalDesign and Outsourcing








